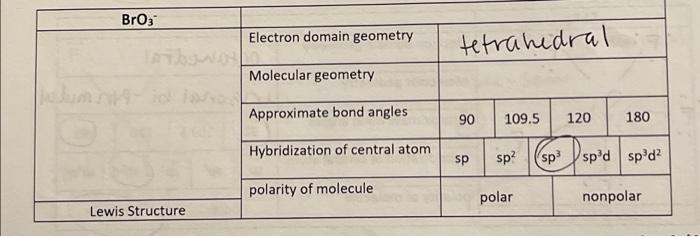
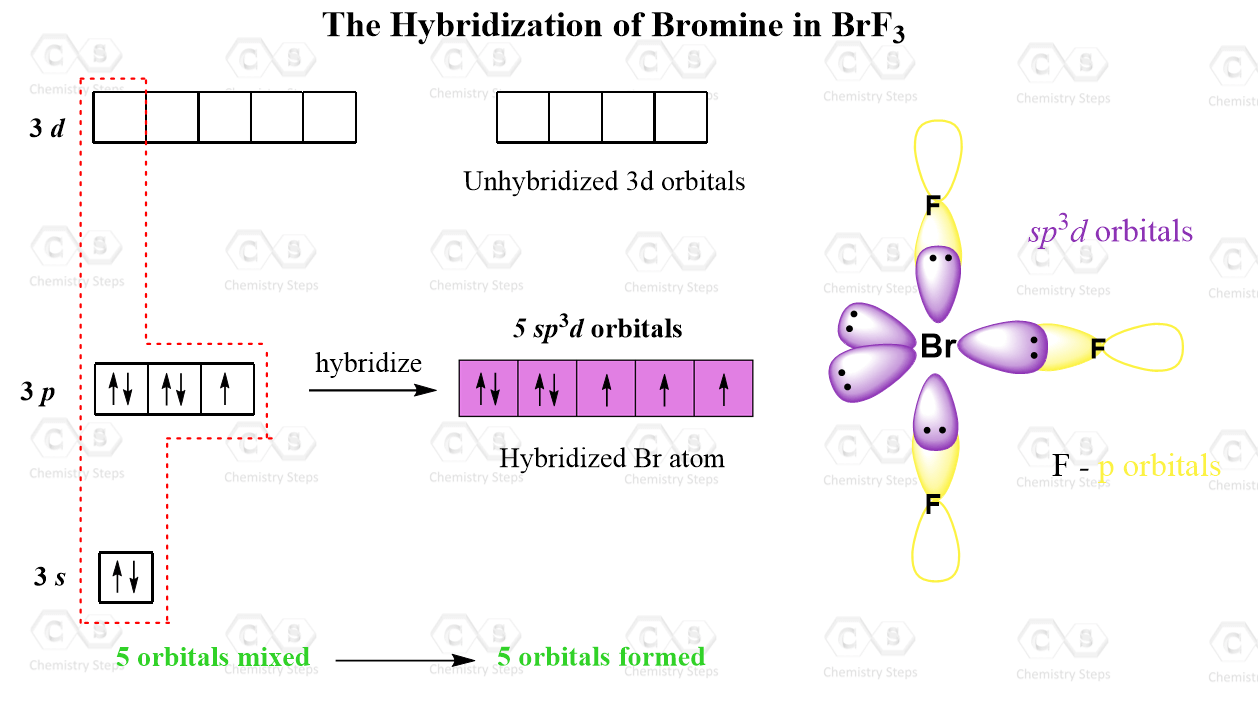
The hybridization and geometry of Br{F}_{3} molecules are:{ sp }^{ 3 }{ d }^{ 2 } and tetragonal{ sp }^{ 3 }d and T-shaped{ sp }^{ 3 }d and bentnone of these

SOLVED: Question 9 (10 points) What is the hybridization of the central atom in AsBr32- (OR AsBr3 2-)? sp sp² sp³ sp³d sp³d² Question 10 (10 points) What is the hybridization of

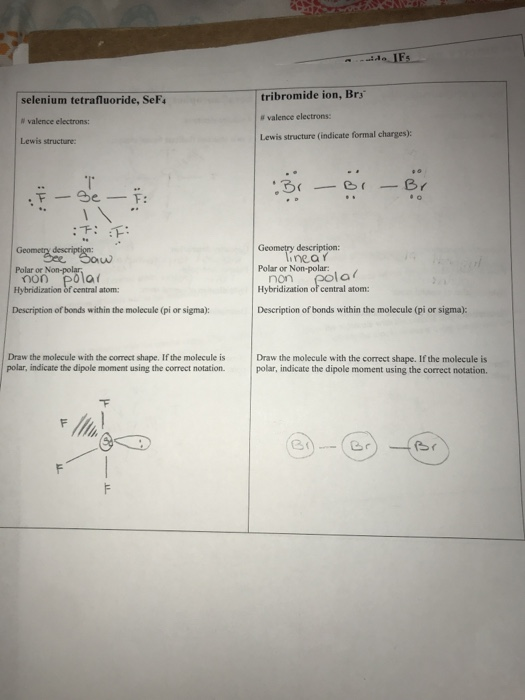
While I3- and Br3- are both stable molecules, F3- is not a stable molecule. Provide an explanation. | Homework.Study.com
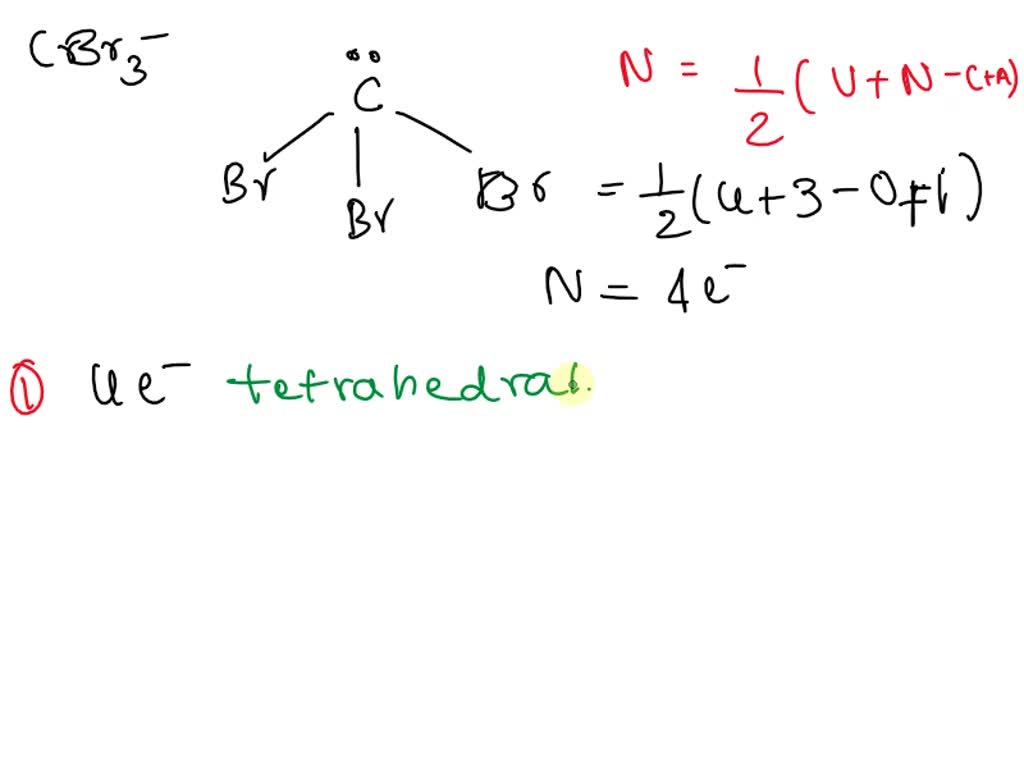
SOLVED: Give the electron geometry, molecular geometry, and hybridization for CBr3-. Electron geometry: trigonal planar Molecular geometry: trigonal pyramidal Hybridization: sp3

SOLVED: Based on hybridization, what is the polarity of Br3? A) Non-polar B) Ionic C) It cannot be determined. D) Polar Based on hybridization, what is the polarity of Brs? A) Non-polar

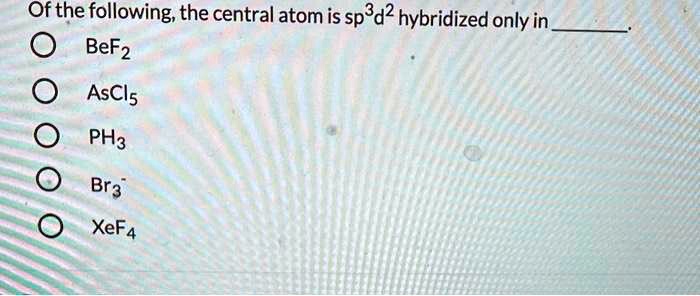
SOLVED: Of the following, the central atom is sp3d2 hybridized only in BeF2, AsCl3, PH3, Br3, O, and XeF4.

give the structure and mean rion the hybridisation of central atom of the following (a)if7 (b) brf3 (c) - Brainly.in