
Which of the following compounds exhibit d2sp3 hybridization? Select "yes" for molecules with d2sp3 hybridization and "no" for all others. KrCl4 TeF6 PBr5 KrCl2 ICl5 IF3 TeCl4 | Homework.Study.com

A complex involving d^2sp^{3} hybridization is :a square planar geometrya tetrahedral geometryan octahedral geometrytrigonal planar geometry
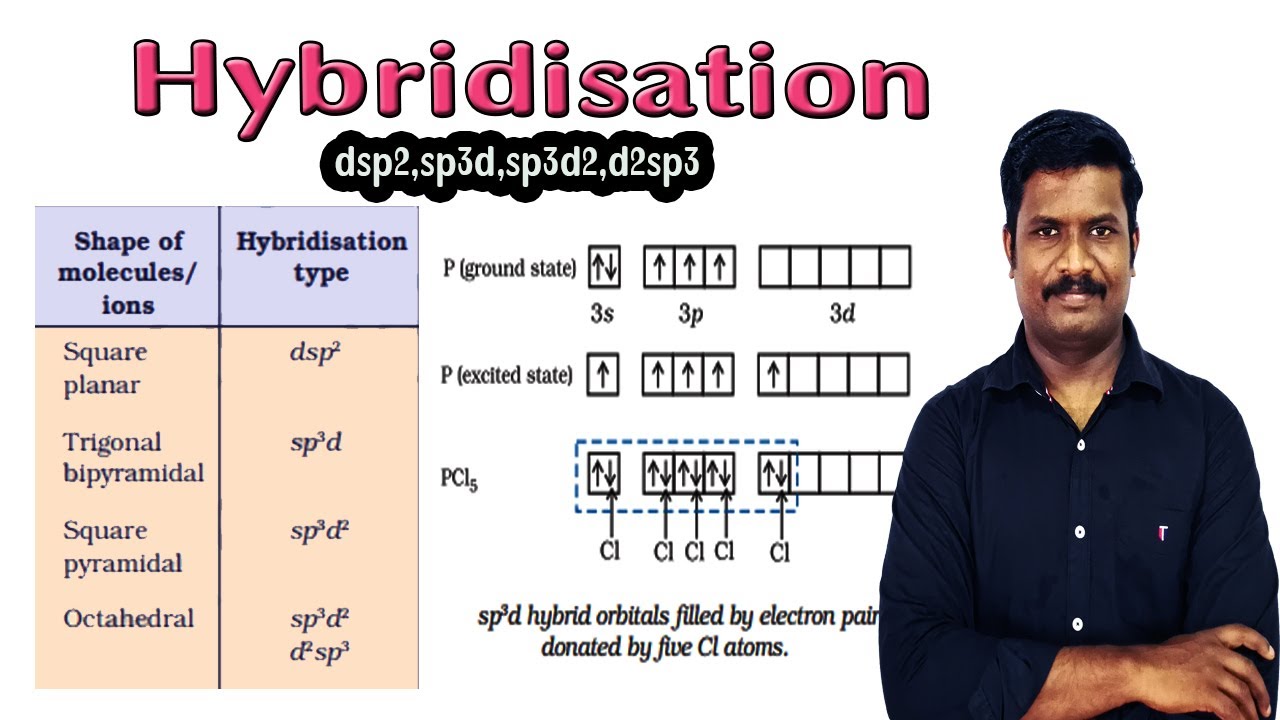
10.Hybridisation | dsp2 | sp3d | sp3d2 | | d2sp3 | Hybridisation Involving d Orbitals | ncert - YouTube
Difference Between sp3d2 and d2sp3 Hybridization Key Difference - sp3d2 vs d2sp3 Hybridization What is sp3d2 Hybridization

Odes it make any difference if we write hybridisation as sp3d2 or d2sp3 Shape of molecules/ ions Square planar - Chemistry - Chemical Bonding and Molecular Structure - 13299455 | Meritnation.com


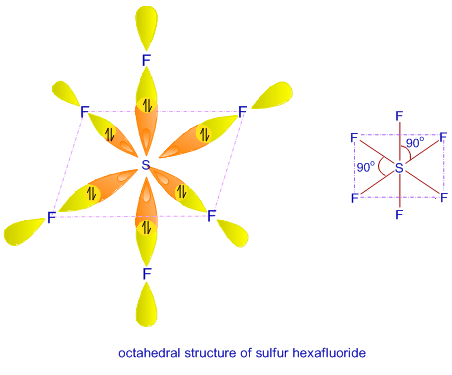




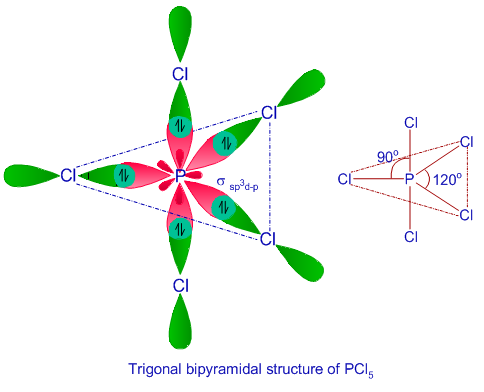
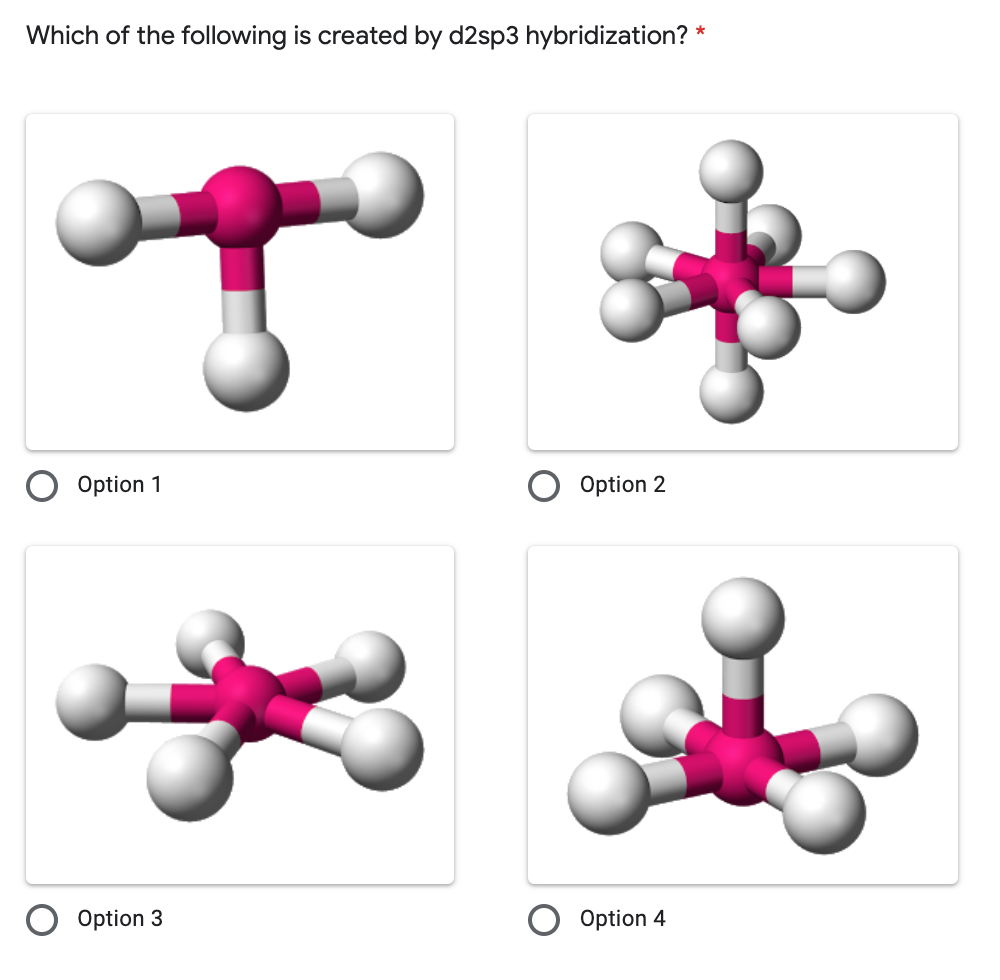





![Hybridisation of [Ni(NH3) 6]2+ is d2sp3 or not?????? - EduRev NEET Question Hybridisation of [Ni(NH3) 6]2+ is d2sp3 or not?????? - EduRev NEET Question](https://edurev.gumlet.io/ApplicationImages/Temp/5610672_cf5cdd6f-a6a7-441d-a688-265da1aa183a_lg.png)