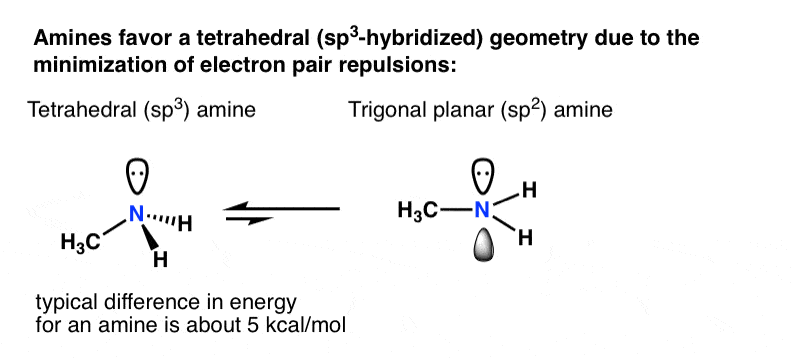
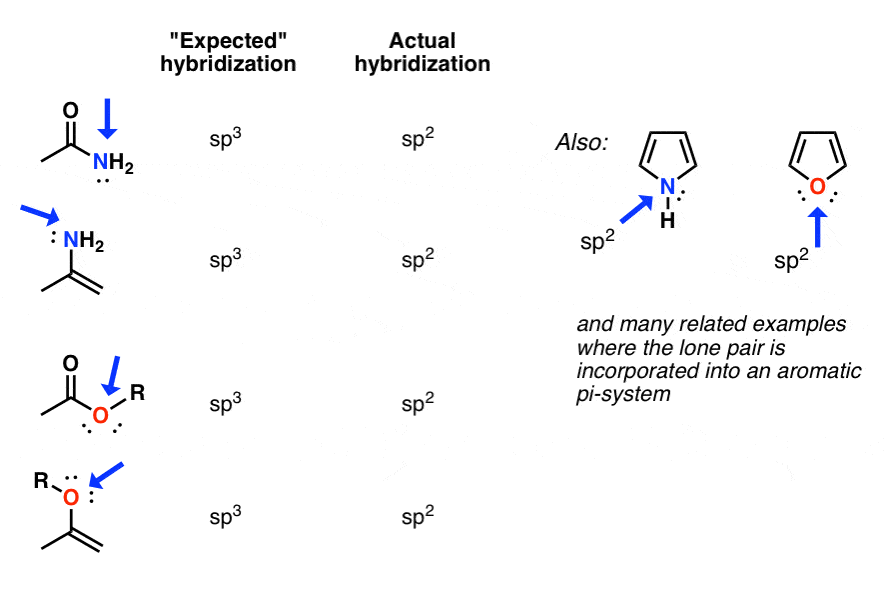
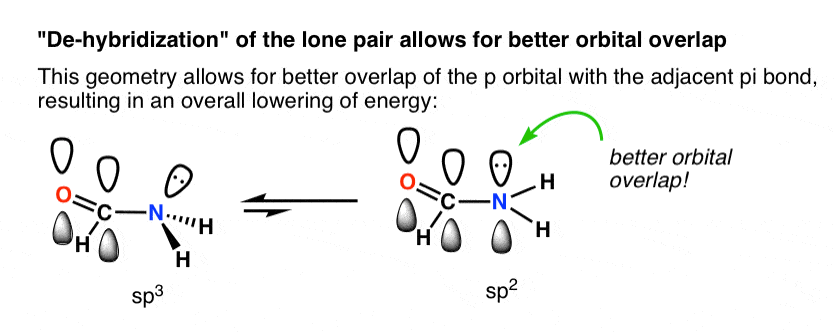
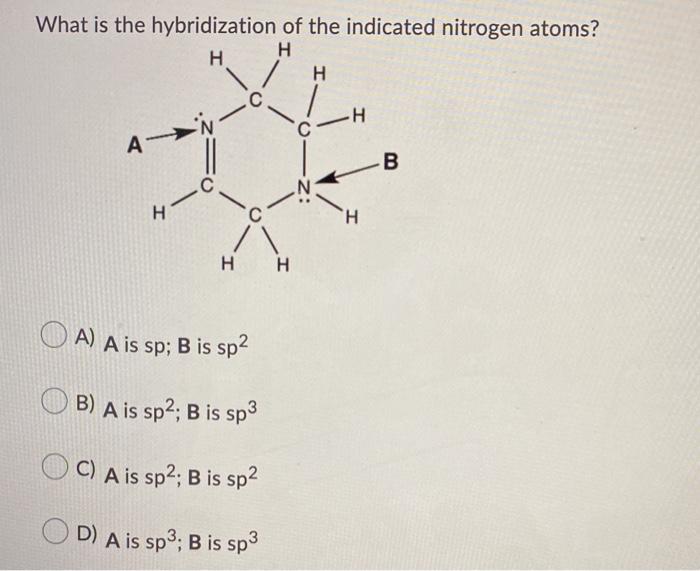
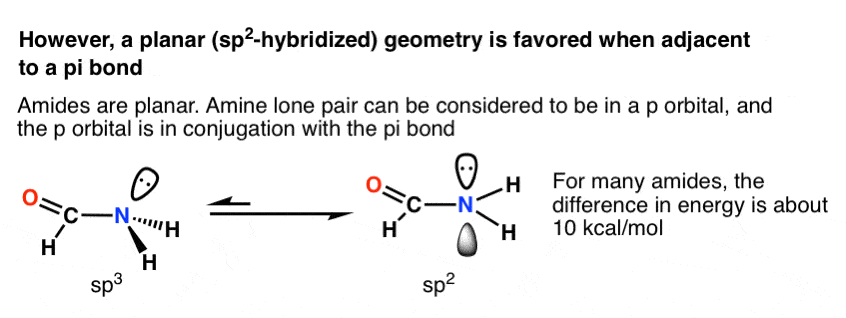
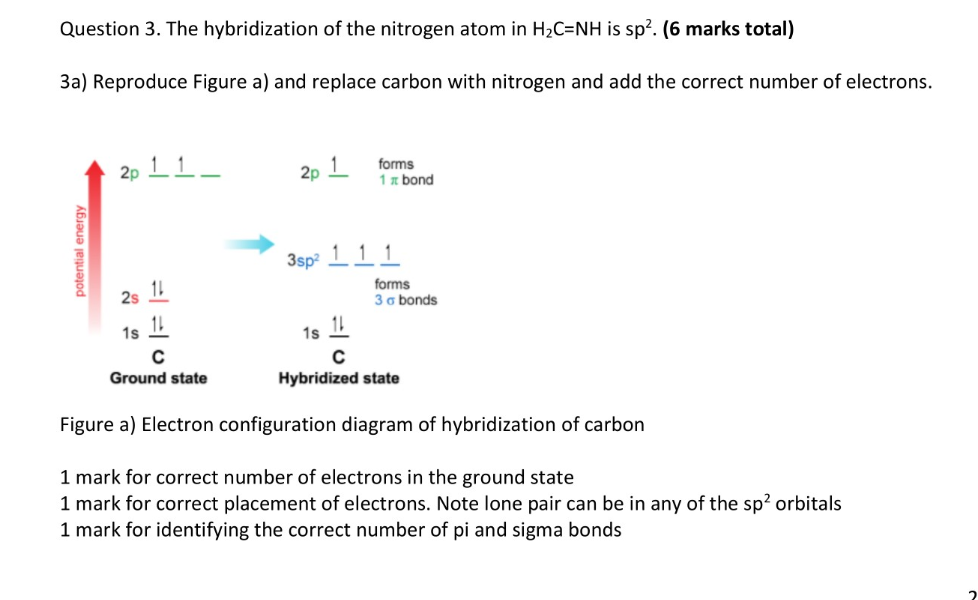
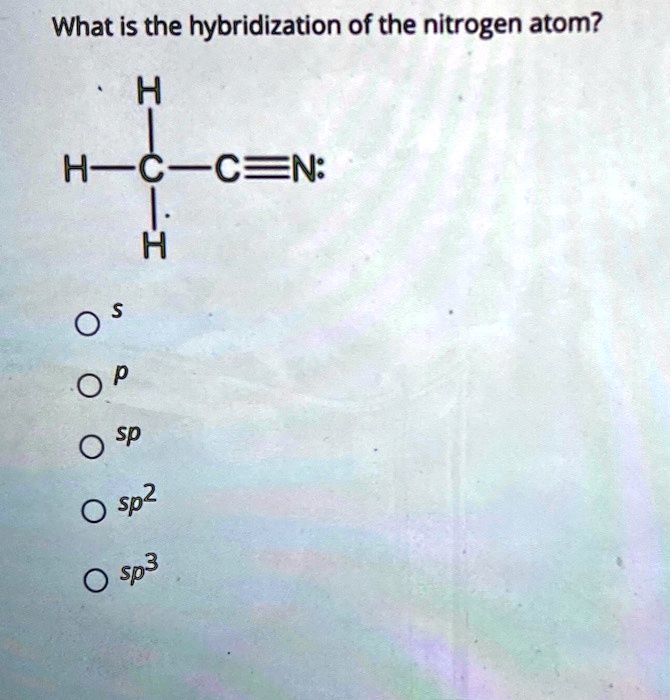
Experimental evidence suggests that the nitrogen atom in ammonia, NH3, has four identical orbitals in the shape of a pyramid or tetrahedron. Draw an energy-level diagram to show the formation of these

NO2 Hybridization (Nitrogen Dioxide) | NO2 Hybridization (Nitrogen Dioxide) Nitrogen Dioxide or Nitrogen oxide is a molecule that consists of one Nitrogen and two Oxygen atoms. In this video... | By Geometry

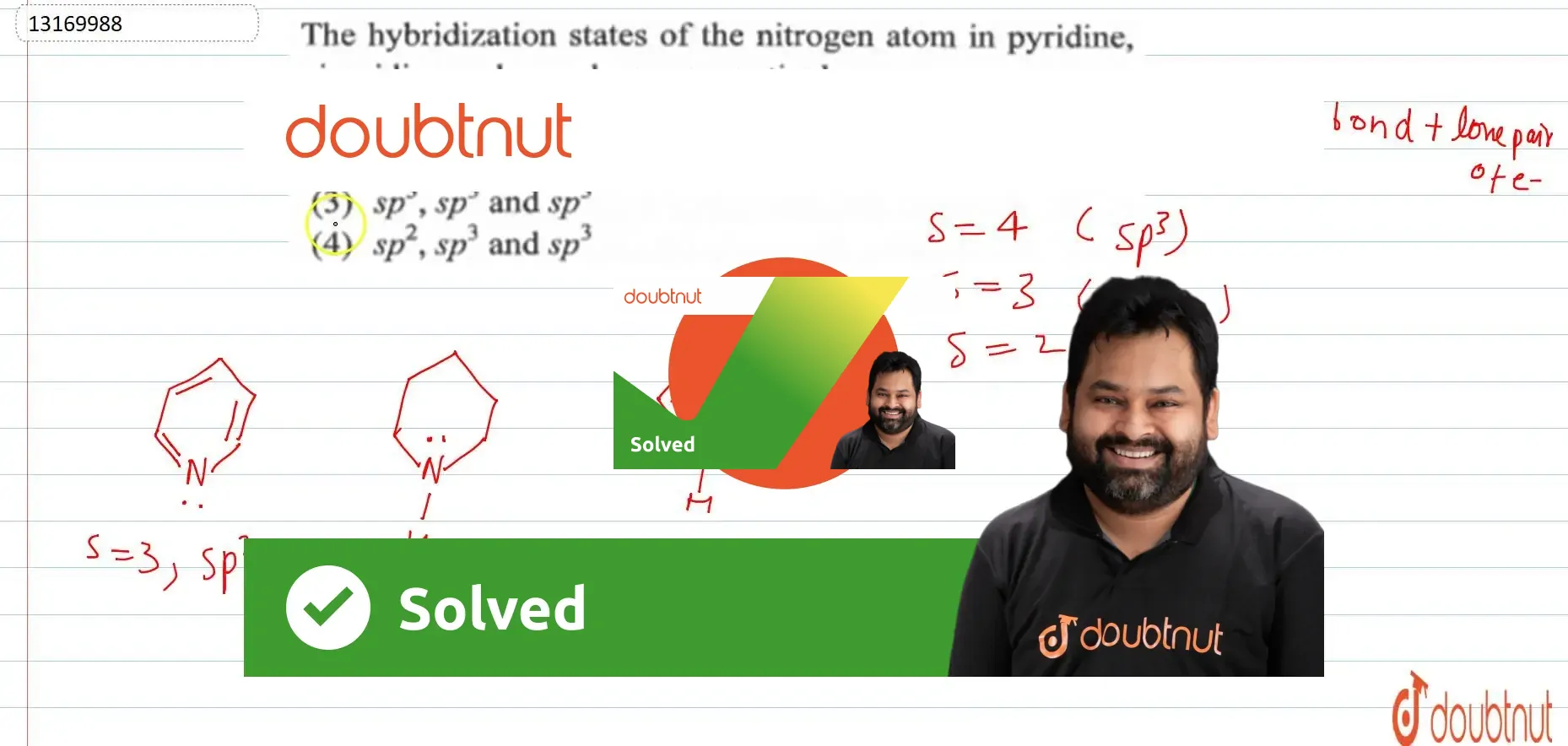
The hybridization states of the nitrogen atom in pyridine piperidine and pyrrole are respectively - YouTube