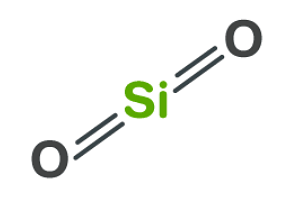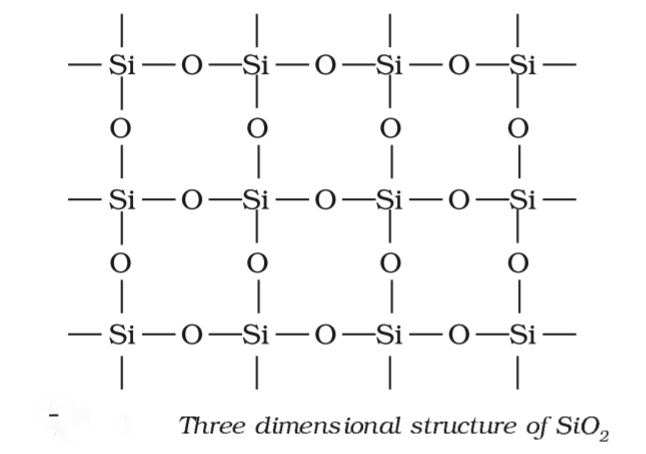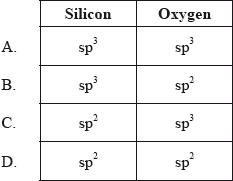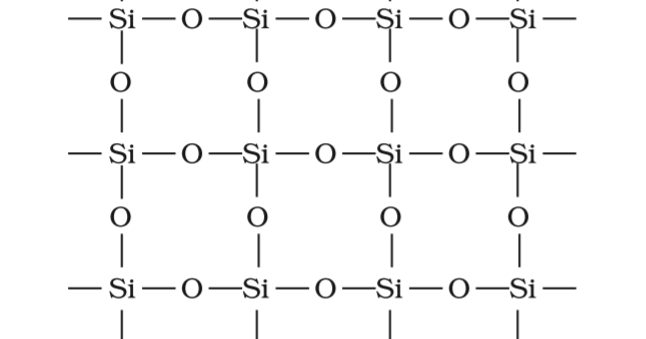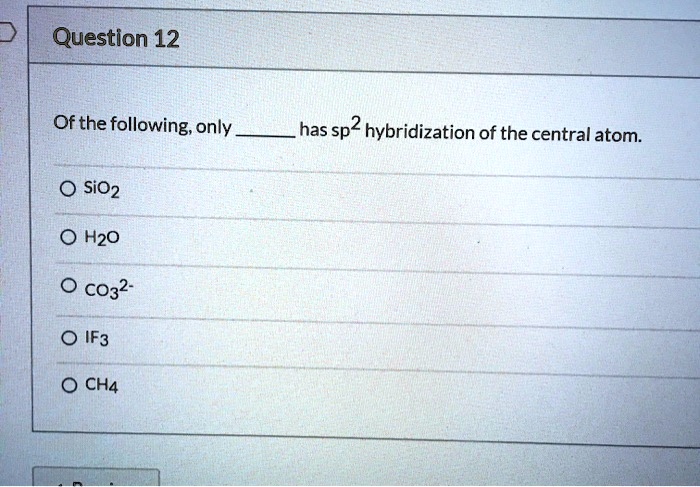
SOLVED: Question 12 Of the following, only has sp2 hybridization of the central atom: SiO2 H2O CO3^2- IF3 CH4

SiO_2 has Two oxygen atoms,each bonded with double bond to SiFour oxygen atoms around Si in same planeFour oxygen atoms bonded to Si tetrahedrallyThree oxygen atoms around each Si in triangular
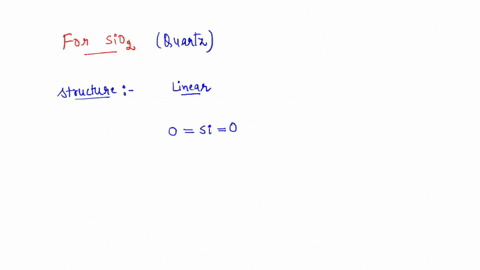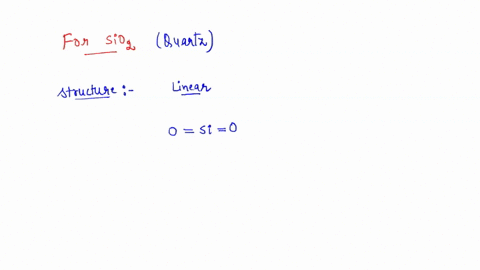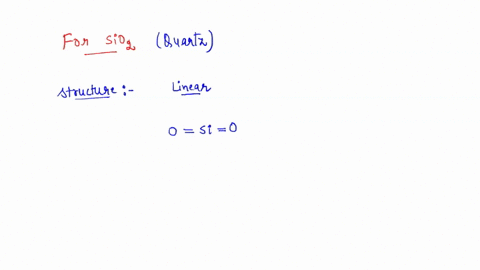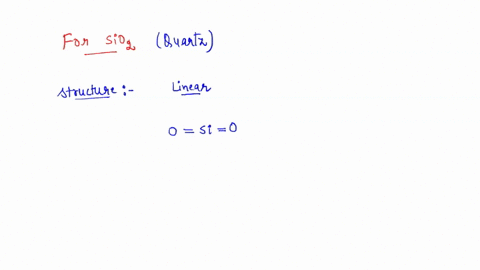
SOLVED: the hybridization of SiO2 identify the number of sigma and pi bonds, and draw the structure to show those bonds and intermolecular forces (identify the type).
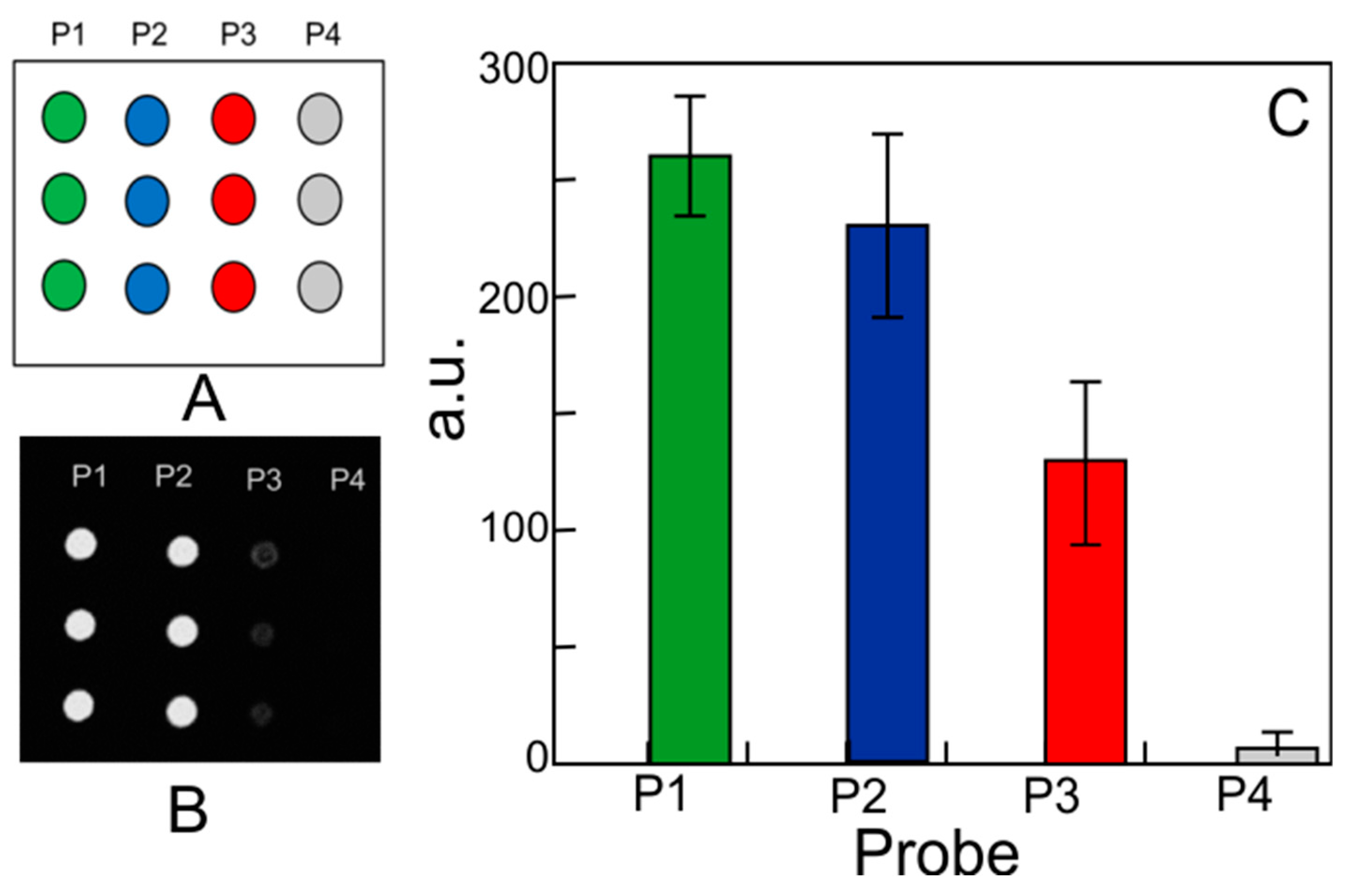
Chemosensors | Free Full-Text | Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations

Carbon–SiO2 Hybrid Nanoparticles with Enhanced Radical Stabilization and Biocide Activity | ACS Applied Nano Materials
![What is the hybridization of the central atom of $SiO_2$?A.\\[sp\\]B.\\[s{p^2}\\]C.\\[s{p^3}\\]D.\\[s{p^3}d\\] What is the hybridization of the central atom of $SiO_2$?A.\\[sp\\]B.\\[s{p^2}\\]C.\\[s{p^3}\\]D.\\[s{p^3}d\\]](https://www.vedantu.com/question-sets/bfbe5949-20d8-4df4-b2df-ad4de6f9d4df3600507102077243315.png)