Gastric Cancer Foundation FDA Grants Priority Review to TAS-102 for Gastric/GEJ Cancer | Gastric Cancer Foundation

Usefulness of TAS-102 as Third-line Chemotherapy for Metastatic Colorectal Cancer | Anticancer Research

Adherence, Dosing, and Managing Toxicities With Trifluridine/Tipiracil (TAS‐ 102) | Semantic Scholar

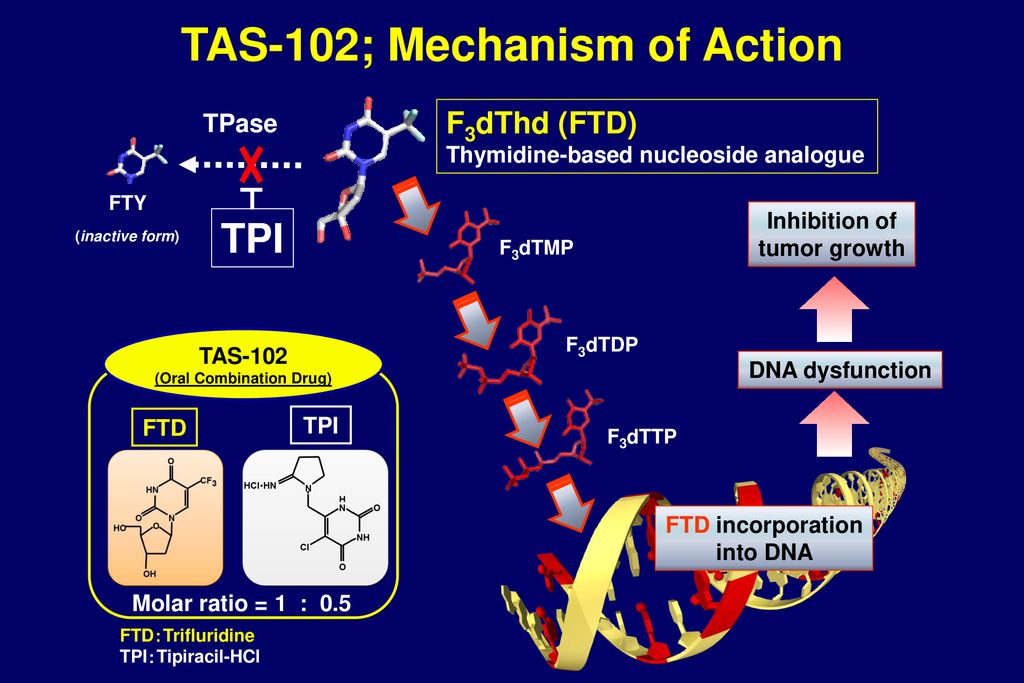
Mode of action of TAS-102. α,α,α-Trifluorothymidine (FTD) is converted... | Download Scientific Diagram
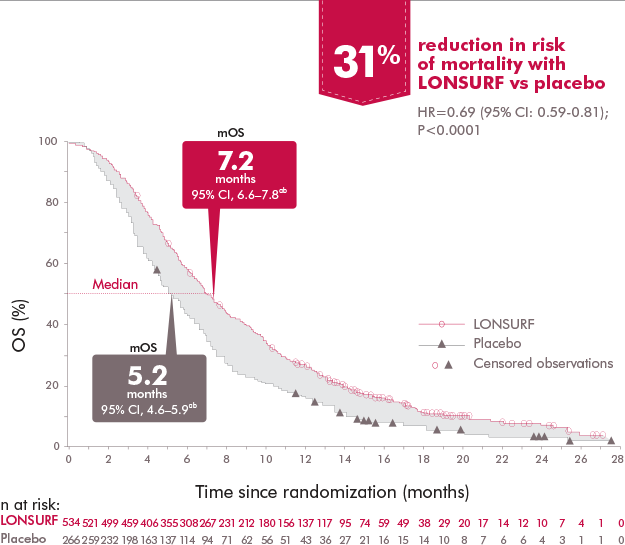
LONSURF® (trifluridine and tipiracil) tablets | Previously Treated mCRC Treatment | TAS-102 Efficacy
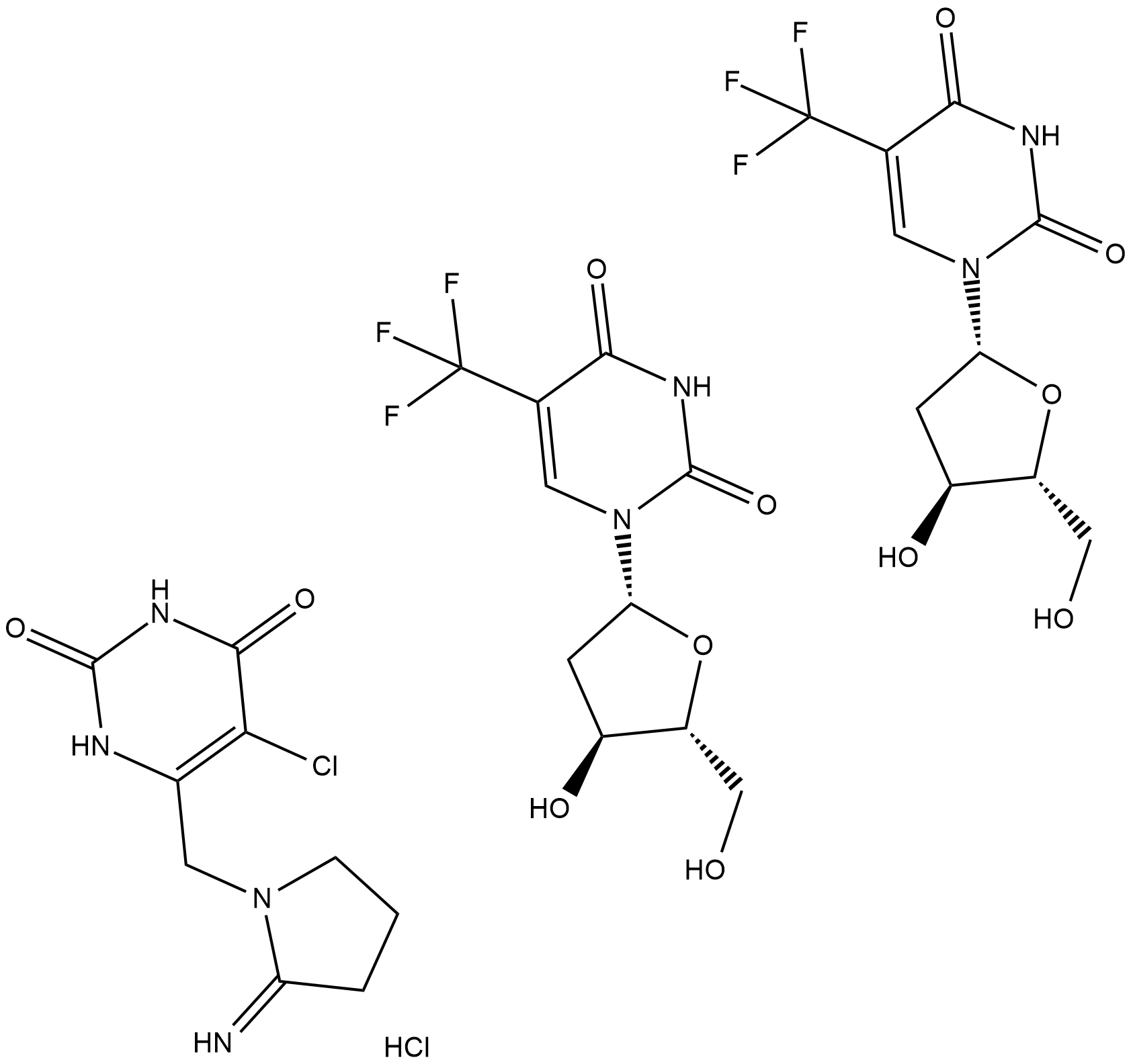
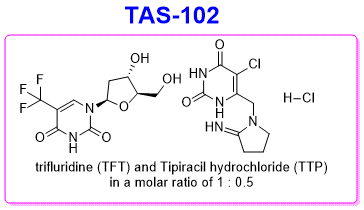
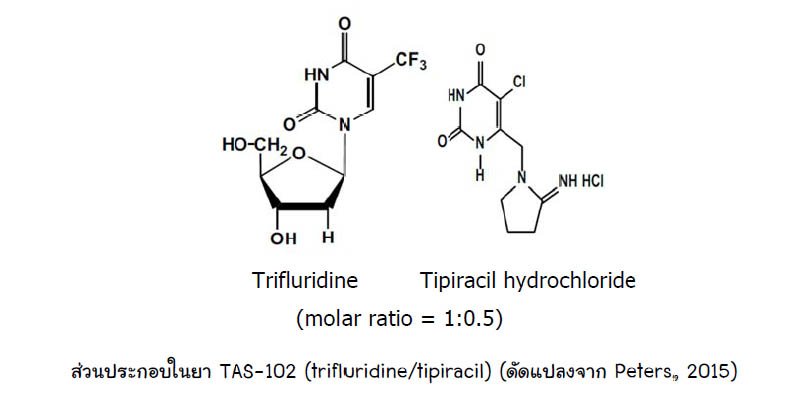
TAS-102 (Trifluridine-tipiracil hydrochloride mixture) | Nucleoside Antimetabolite/Analog Inhibitor | Cas# 733030-01-8 - GlpBio

TAS-102 monotherapy for pretreated metastatic colorectal cancer: a double-blind, randomised, placebo-controlled phase 2 trial - The Lancet Oncology
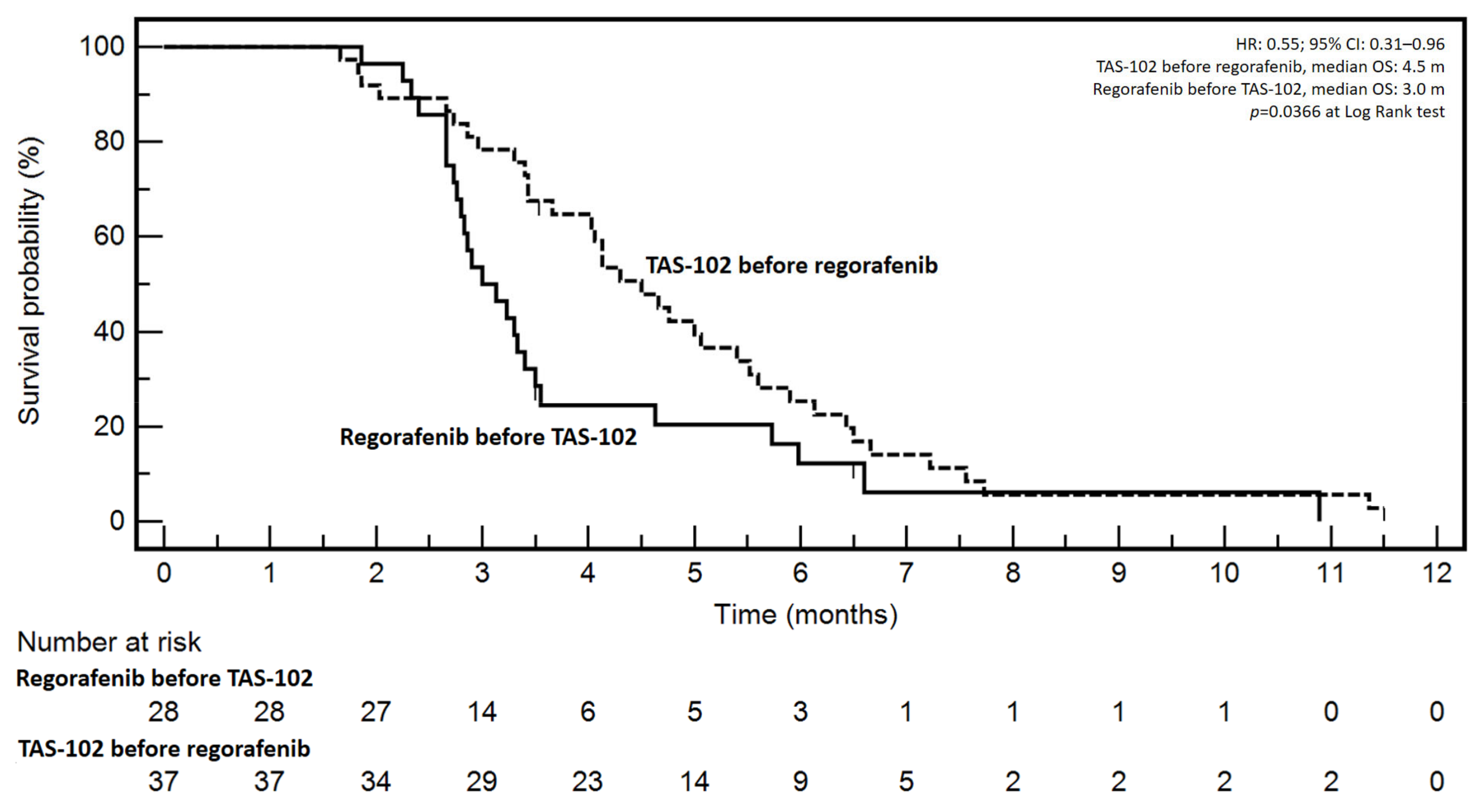
JCM | Free Full-Text | Survival and Toxicities of Metastatic Colorectal Cancer Patients Treated with Regorafenib before TAS-102 or Vice Versa: A Mono-Institutional Real-Practice Study

SUNLIGHT: Phase III Trial of TAS-102 With or Without Bevacizumab for Third-line Treatment of Refractory Metastatic Colorectal Cancer | CCO

Prospective Multicenter Phase II Study of Biweekly TAS-102 and Bevacizumab for Metastatic Colorectal Cancer | Anticancer Research